Website designed by: Vanmala VONGKINGKEO
Icons made by Twitter from www.flaticon.com
Below is a definition of Diabetic Cardiomyopathy by the Dr. Miguel Rodriguez-Ramos.
Diabetes is a major risk factor for heart disease.
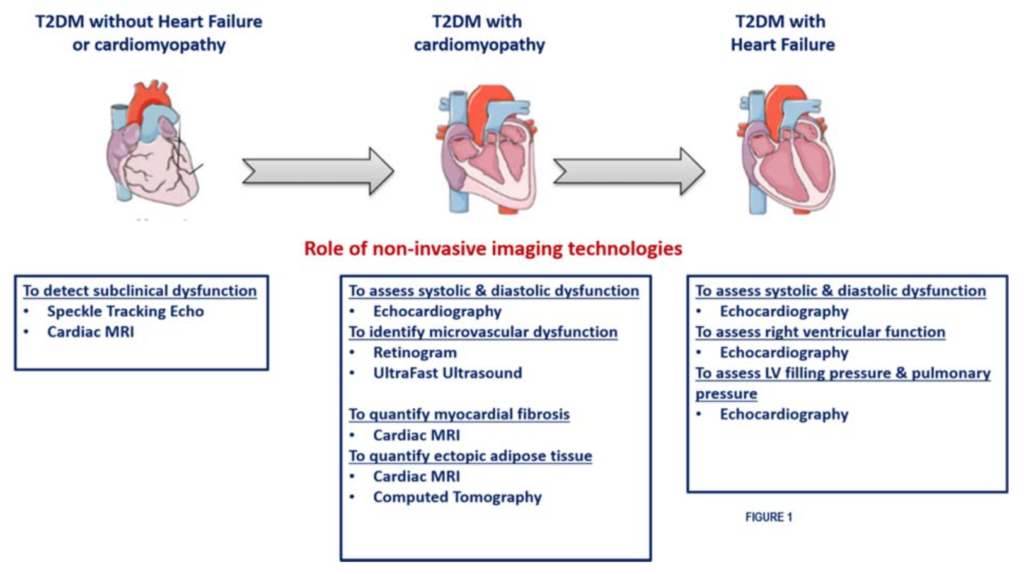
Diabetic cardiomyopathy is a long-lasting process that affects the myocardium in patients who have no other cardiac conditions. The condition has a complex physiopathology which can be subdivided into processes that cause diastolic and/or systolic dysfunction.
It is believed to be more common than reported, but this has not been confirmed by a large study. Diagnosis can involve imaging; biomarkers cannot be used to identify diabetic cardiomyopathy at an early stage.
In people with diabetes, there should be a focus on prevention and, if diabetic cardiomyopathy develops, the objective is to delay disease progression.
Further studies into identifying and managing diabetic cardiomyopathy are essential to reduce the risk of heart failure in people with diabetes.
Doctors diagnose heart disease in diabetes based on
- your symptoms
- your medical and family history
- how likely you are to have heart disease
- a physical exam
- results from tests and procedures
Tests used to monitor your diabetes—A1C, blood pressure, and cholesterol—help your doctor decide whether it is important to do other tests to check your heart health.
A rapidly evolving epidemic in Type 2 Diabetes (T2DM) is afflicting all ages, sexes and socioeconomic classes which includes serious comorbidities such as heart diseases.
While ischaemic heart disease represents the major cause of death of T2DM patients, heart failure (80% of Heart Failure with preserved Ejection Fraction) is the second most common cardiovascular disease in T2DM patients.
The aim of CARDIATEAM is to determine whether T2DM represents a central mechanism contributing to the pathogenesis and progression of a specific cardiomyopathy, called “diabetic cardiomyopathy” (DCM), assessing whether DCM is unique and distinct from the other forms of heart failure.
To achieve this aim CARDIATEAM will build up a deeply phenotyped cohort, including an innovative imaging protocol, based on privileged access within the CARDIATEAM to already existing highly pertinent cohorts of diabetes and heart failure patients and control groups. Central biobanking of the cohort samples will allow detailed omics analysis that will feed together with the phenotype and imaging data into the central CARDIATEAM database. The data gathered will enable unsupervised machine-learning for clustering this heterogeneous population on phenotypic differences beyond diabetes. State-of-the-art big-data processing techniques and disease modeling will allow for controlling for common confounders such as BMI, smoking, age and blood pressure and will finally lead to the identification of new imaging and molecular biomarkers as well as understanding the taxonomy of the development and progression of DCM. Tailored preclinical models will be developed to explore the identified pathways revealing new therapeutic targets.
The results of CARDIATEAM will be able to impact clinical care with the stratification of patients into risk groups of developing DCM, earlier diagnosis of DCM and an improvement of therapy thanks to better assessment of underlying pathophysiology and identification of new biomarkers.

Provide access to cohorts within CARDIATEAM consortium for

Explore by unbiased statistical clustering analysis the specific contribution of Type 2 Diabetes to the development of diabetic cardiomyopathy.

Explore the role of confounding factors that include ageing process and metabolic exposome in inducing diabetic cardiomyopathy.

Understand the physiological pathways responsible for establishment and progression of diabetic cardiomyopathy by translating bedside phenotypic data to bench preclinical models of diabetic cardiomyopathy.

Use diabetic cardiomyopathy as a representative comorbidity for the paradigm of Heart Failure and a targetable mechanism for proof-of-concept studies

Explore the role of existing and newly identified biomarkers for risk stratification of Type 2 Diabetes patients and their potential application for personalised preventive and therapeutic strategies.
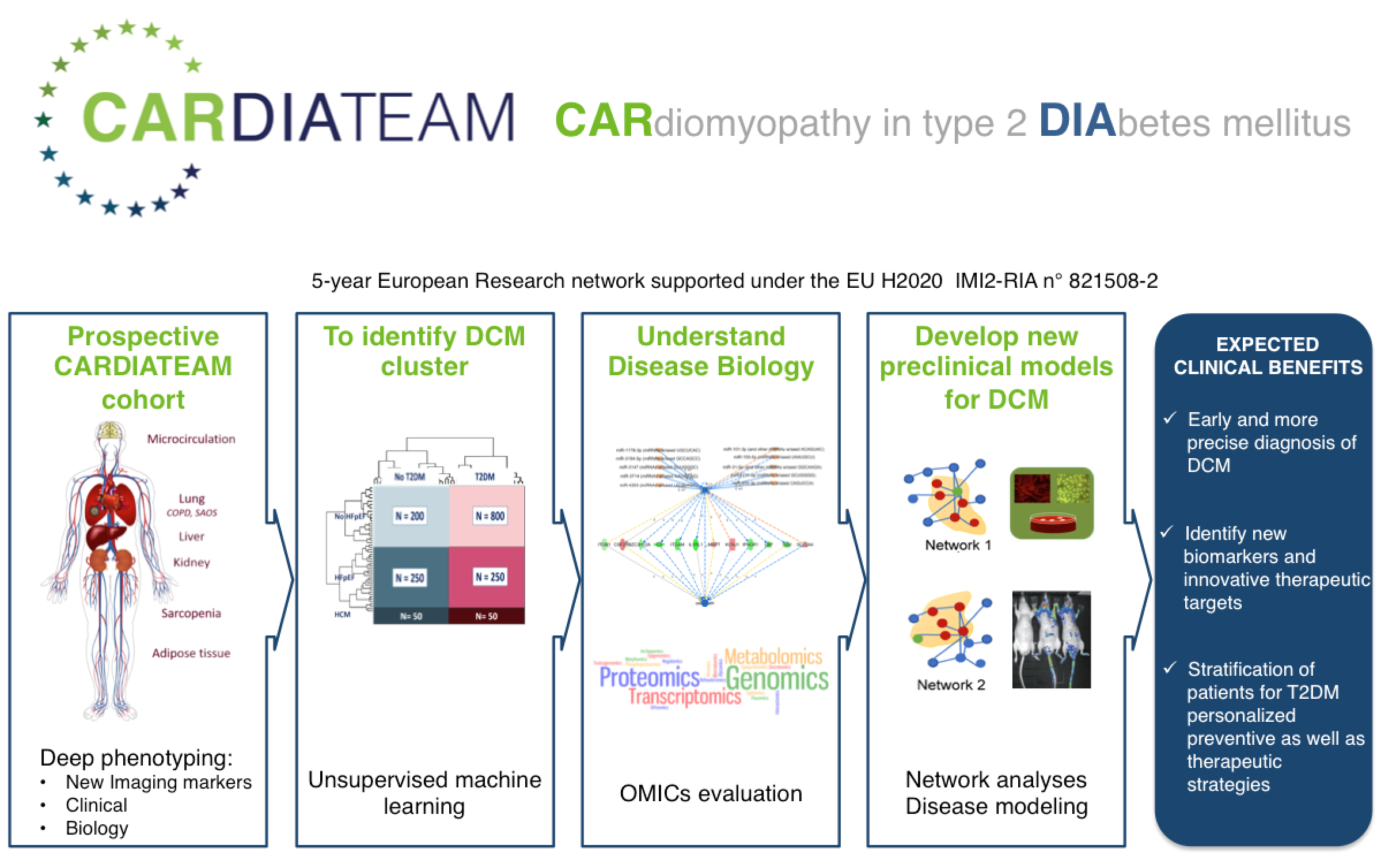
CARDIATEAM is a 5-year program consisting of 6 scientific work packages (WPs) that will foster the dynamic of translational research from patient cohorts to preclinical models and the dissemination on the knowledge on DCM to physicians, patients, regulators & health agencies.
Indeed CARDIATEAM consortium is aware of the need of a homogeneous and deep phenotype among patients in order to optimize the identification of the cluster corresponding to DCM patients by unsupervised analysis.
CARDIATEAM will address the uniqueness of DCM and its progression towards HFpEF by:
the exploitation of published data from 13 existing cohorts (epidemiological meta-analysis) with deep cardiometabolic phenotype (imaging techniques) and longitudinal follow-up among whose participants will be invited for a deep “re-phenotyping” in a new CARDIATEAM cohort
a prospective CARDIATEAM cohort will be recruited from these existing 16 cohorts using a defined set of selection criteria and will include T2DM and non-diabetic patients with a large spectrum of demographic, metabolic and cardiac clinical data from the 16 cohorts described above. This will yield a wide range of T2DM- related phenotypes including common confounders such as BMI, smoking, age and blood pressure.
systems biology analysis after integration of clinical phenotype, imaging and multi-OMICs datasets
preclinical studies relying on in silico and cellular models, small and large animal models relevant to DCM3
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 821508.
See more on Cordis portal.
The contents of this website and the view expressed in the news and publications are the sole responsibility of the authors and under no circumstances can be regarded as reflecting the position of the European Union.
Read our privacy policy and legal notice.
Website designed by: Vanmala VONGKINGKEO
Icons made by Twitter from www.flaticon.com